If Opiaters Used in First Trimester Pregnancy Only, Can Baby Have Withdrawal at Birth

Managing infants built-in to mothers who take used opioids during pregnancy
Podcast
Posted: May 11, 2018 | Updated: Oct 3, 2019
Chief author(south)
Thierry Lacaze-Masmonteil, Pat O'Flaherty; Canadian Paediatric Society. Updated by Thierry Lacaze-Masmonteil, Fetus and Newborn Committee
Paediatr Child Health 2020 23(3): 220–226.
Abstract
The incidence of infant opioid withdrawal has grown quickly in many countries, including Canada, in the final decade, presenting pregnant health and early brain development concerns. Increased prenatal exposure to opioids reflects rising prescription opioid employ as well as the presence of both illegal opiates and opioid-commutation therapies. Infants are at loftier chance for experiencing symptoms of abstinence or withdrawal that may crave assessment and treatment. This practice bespeak focuses specifically on the effect(south) of opioid withdrawal and current management strategies in the care of infants built-in to mothers with opioid dependency.
Keywords: Discharge planning; Management; NAS; NPI; Treatment strategies
BACKGROUND
For 2016–17, the Canadian Institute for Health Information reported that an estimated 0.51% of all infants built-in in Canada (approximately 1850/year, Quebec excluded) had Neonatal Abstinence Syndrome (NAS). A large pct of these cases are attributed to opioid withdrawal [i]. The average length of stay in acute intendance facilities for these infants was xv days. Contempo reports indicate the number of infants requiring ascertainment for withdrawal symptoms is increasing annually and that cases are generally under-reported [2]. The costs of hospitalization speak to the significant brunt this problem places on the health of mothers, infants and families, forth with infirmary units, wellness care providers and other community resources [3].
Opioid use during pregnancy, whether prescribed or illicit, can be associated with negative pregnancy and infant outcomes, including prematurity, low nascency weight, increased risk of spontaneous abortion, sudden infant death syndrome and baby neurobehavioural abnormalities [four]. Natural compounds include morphine and codeine. Heroin, oxycodone, hydrocodone, hydromorphone and buprenorphine are semi-constructed. Fentanyl, methadone, normethadone, tramadol and meperidine are synthetic [5]. The most significant furnishings of maternal opioid dependence and prenatal fetal exposure are short-term complications, with NAS predominating. NAS is a set of drug withdrawal symptoms that can bear on the cardinal nervous system (CNS), and the gastrointestinal and respiratory systems in the newborn [half dozen]. Potential long-term outcomes of prenatal opiate exposure are difficult to predict due to multiple, interrelated variables of maternal-infant chance factors that are known to impact developmental outcomes in this cohort [7][eight].
Meaning women with an opioid dependence should exist brash to continue or commence an opioid maintenance therapy plan [9]. Methadone is often recommended for opioid-dependent pregnant women [x] and some studies suggest buprenorphine every bit an alternative handling [11]. The literature reports that opioid substitution therapy during pregnancy may lessen the use of other opioids and illicit drugs and ameliorate prenatal care, including access to education, counselling and customs supportive services [12][thirteen]. Obtaining a comprehensive medication or illicit substance use history and a focused social history are recommended. Other associated health risks include infections (east.m., hepatitis B, hepatitis C, syphilis and HIV), insufficient maternal nutrition or admission to antenatal care, too every bit social adventure factors, and should be screened for, adamant and managed appropriately.
CLINICAL PRESENTATION
The presentation of withdrawal symptoms varies, depending on the type of maternal opioid used, the frequency, dose and timing of last exposure, gestational age, maternal metabolism and maternal utilize of other substances. Approximately 50% to 75% of infants born to women on opioids will crave treatment for opioid withdrawal symptoms [14]. Symptoms of opioid withdrawal (Figure one) typically appear before long after birth, with the majority exhibited inside the kickoff 48 h to 72 h postdelivery. Some reports suggest a later symptom presentation, at v to 7 days postbirth, afterwards exposure to methadone or buprenorphine [xv][16]. Initial acute symptoms may persist for several weeks (ranging from 10 to xxx days) while milder symptoms, such equally irritability, sleep disorders and feeding problems can persist for 4 to 6 months [17]. Preterm infants have been described as being at lower risk for drug withdrawal and symptoms of NAS may not exist as apparent as in their term counterparts [eighteen][19]. Reasons for this difference include: a shorter in utero exposure time, decreased placental manual, inability to fully excrete drugs by immature kidneys and liver, minimal fat stores leading to lower opioid degradation and activity, likewise as a limited chapters to express classic NAS symptoms by the immature brain [19].
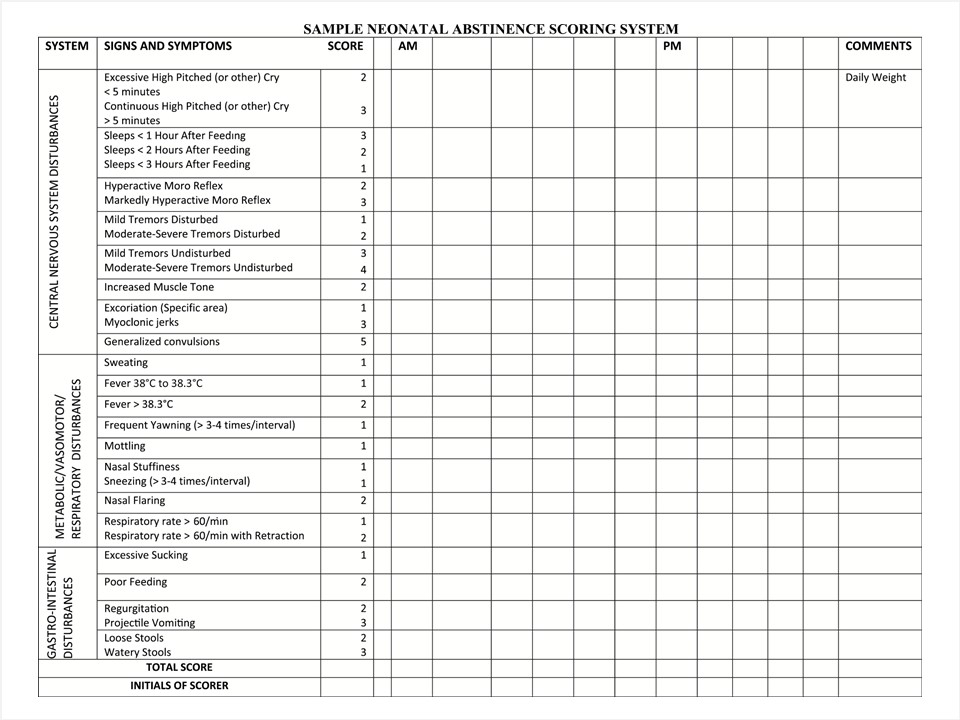 |
| Effigy 1. Modified Finnegan scoring system. Reproduced from refs. [15][twenty][21]. |
Other neonatal conditions such as hypoglycemia, hypocalcemia, CNS injury, hyperthyroidism, bacterial sepsis or other infections may present with like symptoms and should be considered as part of the differential diagnosis.
ASSESSMENT
For all opioid-exposed infants, an assessment scoring organisation should be used to measure the severity of withdrawal symptoms and help make up one's mind whether boosted monitoring, nursing, medical and pharmacological therapy are required [20]. The near widely used scale to evaluate infants for opioid withdrawal is the Finnegan scoring system, which identifies behaviours associated with withdrawal (Figure 1) [21]. An initial score is typically obtained inside the first 1 h to ii h postdelivery, then rechecked every 3 h to four h thereafter, in conjunction with other nursing assessments. Nearly guidelines suggest that scoring should be done for a minimum of 72 h and up to 120 h when an baby was exposed in utero to a longer-acting opioid, such as methadone or buprenorphine [19]. The scale may also be used to appraise the resolution of symptoms afterward initiating treatment. Accurate, consequent scoring of symptoms is necessary to ensure that the infant receives appropriate care [22]. A variety of instructional videos and "train-the-trainer" approaches are available to ensure that staff education is consistent for proper scoring [23]. Priorities are to develop a standardized process to identify, manage and evaluate infants with NAS, and so to discharge them as soon as it is condom and appropriate.
MANAGEMENT
Successful supportive direction of infants exposed to opioids during pregnancy depends on a number of factors, such as providing appropriate medication(due south), correct scheduling, using an accurate tool to measure and evaluate the severity of symptoms, creating a compatible concrete environment and having a knowledgeable, experienced health care team. Involving interprofessional team members, (i.east., specialized in nursing, neonatal medicine, social work, pharmacy, diet and customs resources) is essential for ensuring the seamless direction and discharge of these vulnerable infants [15]. Treatment goals include preventing complications associated with NAS and restoring normal newborn activities, such every bit sleep, sufficient feeding, weight gain and environmental accommodation.
In addition to maternal cocky-reporting, some practitioners behave urine or meconium toxicology screens for infants born to women with a suspected history of drug abuse during pregnancy. This data may be helpful to place the substance(s) of abuse and for selecting appropriate pharmacological treatment, when required [16]. However, the validity and reliability of toxicology testing is controversial [24]. Further, obtaining maternal consent for testing, securing knowledge of who will accept access to the results and other legal considerations are important in the decision to comport or send toxicology screens [25]. National neonatal resuscitation guidelines suggest that using naloxone during infant resuscitation should exist avoided when an baby is born to a known opioid-dependent female parent because it has been associated with seizures in newborns [26]. There is no show to suggest that a higher than routine level of wellness care practitioner (HCP) care competency is required at delivery, provided in that location are no other chance factors present.
Information technology is well known that separating babe and mother can be detrimental to early attachment. Contempo literature supports practices that go on opioid-dependent mothers and their infants together from birth, such as rooming-in. Such practices accept additional benefits, such every bit lower neonatal intensive intendance unit of measurement (NICU) admissions, higher breastfeeding initiation rates, less demand for pharmacotherapy and shorter infirmary stays [27]–[30]. A rooming-in model of care—rather than admission to NICU—tin be considered for mother–infant dyads at take chances for developing NAS symptoms when: infants are term or well-nigh term, medically stable, and adequate resources are in place to support both the family and HCPs.
Nonpharmacological interventions
Initial handling for neonatal withdrawal should exist primarily supportive considering medical interventions tin can prolong hospitalization, disrupt mother–infant zipper and subject an infant to drugs that may not exist necessary. Nonpharmacological interventions have been shown to reduce the effects of withdrawal and should be implemented as shortly as possible following birth [two]. Examples of supportive interventions include skin-to-skin contact, safe swaddling, gentle waking, tranquility environs, minimal stimulation, lower lighting, developmental positioning, music or massage therapy [31].
Breastfeeding should be encouraged because it tin can delay the onset and subtract the severity of withdrawal symptoms besides as decrease the need for pharmacological treatment [32]. HIV-negative mothers who are stable and on opioid maintenance treatment with either methadone or buprenorphine should be encouraged to breastfeed [ix]. Breastfeeding provides optimal nutrition, promotes maternal–infant attachment and facilitates parenting competence. Mothers with a dependency who wish to breastfeed may crave extra support every bit they are less probable to initiate breastfeeding successfully and more likely to stop breastfeeding early [33].
NAS infants may show impaired feeding behaviours such as excessive not-nutritive sucking, poor feeding, regurgitation and diarrhea [34]. An early study [35] found that opioid-exposed infants had more feeding problems (rejecting the nipple, dribbling milk, hiccoughing, spitting up, and coughing) than nondrug-exposed infants. More than recent studies take confirmed these findings and described the challenges caregivers face when feeding infants who show signs of withdrawal [36]. Supplementation with concentrate to increase caloric intake or total fluid intake has been suggested for infants with poor weight gain [37].
Pharmacological interventions
Pharmacological therapy is indicated for infants whose withdrawal signs are increasingly astringent or whose concurrent NAS scores climb despite supportive measures to reduce and manage symptoms. Infants who require treatment with medications may also require admission to a special care nursery or NICU for cardiorespiratory monitoring and observation while therapy is initiated, peculiarly if they are medically unstable. A stabilized baby tin be transferred back into a intendance-past-parent area (e.g., rooming-in) provided that cess, parent education and medication weaning monitoring are ongoing, infant–mother attachment is supported and comprehensive belch planning can be initiated [29]. Rooming-in with mothers on a methadone program and breastfeeding have been shown to reduce the need for pharmacological intervention [30].
Several pharmacological agents have been used to ameliorate symptoms associated with neonatal opioid withdrawal (Table one). Few studies have examined pharmacotherapy efficacy. The American Academy of Pediatrics recommends matching drug option to the type of agent causing withdrawal [19]. Morphine and methadone remain the most common first-line medications, with lack of bear witness for which amanuensis is superior [38]. Compared with oral morphine, sublingual buprenorphine has recently been shown to reduce length of stay in hospital past 42% in symptomatic infants born to mothers on methadone [39]. Adjunctive use of phenobarbital or clonidine therapy to treat opioid withdrawal has been studied and implemented in some guidelines [40][41]. Published guidelines provide information on initial dosing, dosing increments, initiating additional treatments and weaning, to assist in a consistent approach to management [42].
DISCHARGE CONSIDERATIONS
Length of stay in hospital varies depending on prenatal drug(s) exposure, severity of withdrawal, symptoms, treatment and social factors [42]. Observe for a minimum of 72 h. If the treatment threshold is not reached inside that time, the infant becomes eligible for discharge [43]. The key to successful transition home is to ensure continuity of care by an interprofessional team, with anticipatory planning for when the baby meets criteria for discharge.
Individualized belch planning should include appropriate referral to a chief health care provider familiar with pharmacological treatments for opioid withdrawal, nutritional and family supportive resource and infant neurodevelopmental assessment. Communicating with the infant's biological (or where necessary, foster care) family and the chief HCP about the discharge plan and follow-up is essential. In some situations, when adequate medical and social follow-up is available, infants may be discharged habitation on pharmacological back up [44][45]. At that place are a few studies, for a select group of infants, which have demonstrated that infants tin can be managed safely in an outpatient setting without increasing handling time [46]. Benefits of home-based detoxification include: decreasing length of hospital stay and associated wellness care costs, promoting infant–flagman attachment and increasing breastfeeding rates [47]. Of course, these benefits must exist weighed confronting possible risks. Before discharge, the babe should exist demonstrating tolerance of pharmacological tapering, with consequent withdrawal scores < 8. Also, a articulate, documented medications weaning plan should be in place for the HCP and family to follow. Other community referrals to consider may include ongoing maternal substance abuse handling programs, public health and child and youth services, a community support worker, infant evolution programs and/or parenting support groups. Also, the families and/or guardians must show they tin can provide a supportive and safe home environment [48].
| Table i. Medications to treat neonatal abstinence syndrome | |||
| Medication | Machinery of action | Dose | Comments |
| Morphine | Natural m -receptor agonist | If score is ≥ 8 on three (or ≥ 12 on 2) consecutive evaluations, beginning at 0.32 mg/kg/day, divided every 4 h–6 h, orally. | Virtually commonly used as first-line handling in Canada |
| Methadone | Synthetic complete m-receptor agonist; N-methyl-D-aspartate receptor adversary | 0.05–0.i mg/kg/dose every 6 h–12 h, orally | Long half-life (26 h) Used in many countries as a first-line treatment (instead of morphine) when mother is on methadone. |
| Phenobarbital | Gamma aminobutyric acid (GABA) receptor agonist | May be used in addition to morphine, especially in poly-substance abuse cases. Loading dose: 10 mg/kg, orally, every 12 h for three doses Maintenance dose: five mg/kg/twenty-four hours, orally. Wean by ten% to twenty% every day or every ii days when symptoms are controlled. | Long one-half-life (45 h–100 h) Requires blood level monitoring May brand GI symptoms worse Sedative effect Contains xv% alcohol |
| Clonidine | Blastoff-2 adrenergic receptor agonist | Culling therapeutic option in combination with morphine. Especially effective when autonomic symptoms of NAS are present. Outset at 0.5 mcg/kg for the first dose. The recommended maintenance dose is 3–v mcg/kg/day divided every iv–six h. Wean by 25% of the total daily dose every other mean solar day (Q4h to Q6h × 48h, to Q8h × 48h, to Q12h × 48h to HS, and so d/c) | Booze-gratuitous grooming available Long half-life (44 h–72 h) Abrupt discontinuation may cause rapid rise in blood pressure (BP) and eye rate (Hr). Gradual weaning is therefore recommended. |
| Buprenorphine | Semi-synthetic partial thousand-receptor agonist, yard-receptor antagonist | four–5 mcg/kg/dose every 8 h; sublingual route Maximum dose 60 mcg/kg/solar day | One-half-life (24 h–60 h) Sublingual assistants of a dilution of buprenorphine solution in ethanol and sucrose Contains 30% booze |
| Data taken from ref. [nineteen]. d/c, discontinue; HS, bedtime. | |||
Summary
Opioid-dependent mothers should exist informed that their baby, who has been exposed to natural, semi-constructed or constructed opioids during pregnancy, will require observation for symptoms and signs of neonatal withdrawal post-obit nascency. Antenatal consultation past perinatology, paediatrics and/or neonatology is encouraged.
All infants at run a risk for developing neonatal opioid withdrawal should exist evaluated using a reliable, valid neonatal withdrawal score instrument.
Data and guidelines for HCPs on managing infants born to opioid-dependent mothers should outline the continuum of intendance. Strategies to support keeping mothers and infants together and breastfeeding are essential. Providing nonpharmacological interventions, such as skin-to-skin contact, developmental positioning, comfort measures, minimizing environmental stimuli, ensuring acceptable diet and providing pharmacological treatment when indicated, are primal components of a comprehensive plan.
An constructive and well-coordinated belch programme that involves an interprofessional wellness intendance team and outlines a comprehensive medications weaning schedule, is essential to ensure seamless transition from hospital to community, and for maintaining continuity of care.
Acknowledgements
This practice point was reviewed by the Community Paediatrics, Drug Therapy and Hazardous Substances and First Nations, Inuit and Métis Health Committees of the Canadian Paediatric Lodge. It has also been reviewed past representatives from the Club of Obstetricians and Gynaecologists of Canada.
CPS FETUS AND NEWBORN Committee
Members: Mireille Guillot Doc, Leonora Hendson MD, Ann Jefferies MD (past Chair), Thierry Lacaze-Masmonteil Doc (Chair), Brigitte Lemyre Doctor, Michael Narvey MD, Leigh Anne Newhook MD (Lath Representative), Vibhuti Shah MD
Liaisons: Radha Chari MD, The Society of Obstetricians and Gynaecologists of Canada; James Cummings MD, Committee on Fetus and Newborn, American Academy of Pediatrics; William Ehman MD, Higher of Family Physicians of Canada; Roxanne Laforge RN, Canadian Perinatal Programs Coalition; Chantal Nelson PhD, Public Health Agency of Canada; Eugene H Ng MD, CPS Neonatal-Perinatal Medicine Section; Doris Sawatzky-Dickson RN, Canadian Association of Neonatal Nurses
Principal authors: Thierry Lacaze-Masmonteil MD, Pat O'Flaherty MEd, MN, RN-EC
Updated by:Thierry Lacaze-Masmonteil MD
References
- Turner SD, Gomes T, Camacho X et al. Neonatal opioid withdrawal and antenatal opioid prescribing. CMAJ Open Enquiry 2015;3(one):E55-61.
- Dow K, Ordean A, Spud-Oikonen J et al.; Neonatal Abstinence Syndrome Work Group. Neonatal abstinence syndrome clinical practice guidelines for Ontario. J Popul Ther Clin Pharmacol 2012;19(3):e488–506.
- Patrick SW, Schumacher RE, Benneyworth BD, Krans EE, McAllister JM, Davis MM. Neonatal forbearance syndrome and associated health care expenditures: United States, 2000-2009. JAMA 2012;307(eighteen):1934–forty.
- Vucinovic Grand, Roje D, Vucinovic Z, Capkun V, Bucat M, Banovic I. Maternal and neonatal effects of substance corruption during pregnancy: Our ten-twelvemonth experience. Yonsei Med J 2008;49(5):705–13.
- Opiate Habit and Treatment Resources. www.opiateaddictionresource.com/opiates/types_of_opioids (Accessed August 22, 2017).
- Finnegan LP. Substance Abuse in Canada: Licit and Illicit Drug Use During Pregnancy; Maternal, Neonatal and Early Babyhood Consequences. Ottawa, Ont.: Canadian Centre on Substance Abuse, 2013: www.ccsa.ca/Resources%20Library/CCSA-Drug-Utilize-during-Pregnancy-Report-2013-en.pdf (Accessed August three, 2017).
- Logan BA, Brownish MS, Hayes MJ. Neonatal abstinence syndrome: Treatment and pediatric outcomes. Clin Obstet Gynecol 2013;56(i):186–92.
- Behnke M, Smith VC; Committee on Substance Abuse; Committee on Fetus and Newborn. Prenatal substance abuse: Curt- and long-term effects on the exposed fetus. Pediatrics 2013;131(3):e1009–24.
- WHO. Guidelines for the Identification and Management of Substance Use and Substance Use Disorders in Pregnancy. Geneva, Switzerland: WHO, 2014. http://apps.who.int/iris/bitstream/10665/107130/one/9789241548731_eng.pdf (Accessed Baronial 3, 2017).
- Wong S, Ordean A, Kahan M; Maternal Fetal Medicine Committee; Family Physicians Advisory Committee; Dr.-legal Committee; Ad hoc Reviewers; Special Contributors. Substance use in pregnancy. J Obstet Gynaecol Can 2011;33(iv):367–84. www.researchgate.net/publication/51059321_Substance_use_in_pregnancy (Accessed Baronial 3, 2017).
- Jones HE, Finnegan LP, Kaltenbach K. Methadone and buprenorphine for the direction of opioid dependence in pregnancy. Drugs 2012;72(6):747–57.
- Ordean A, Kahan M, Graves L, Abrahams R, Boyajian T. Integrated care for pregnant women on methadone maintenance treatment: Canadian primary intendance accomplice report. Can Fam Md 2013;59(10):e462–ix.
- Burns Fifty, Mattick RP, Lim Thousand, Wallace C. Methadone in pregnancy: Treatment memory and neonatal outcomes. Addiction 2007;102(2):264–70.
- Casper T, Arbour M. Testify-based nurse-driven interventions for the care of newborns with neonatal abstinence syndrome. Adv Neonatal Care 2014;fourteen(six):376–80.
- Ordean A, Chisamore BC. Clinical presentation and management of neonatal abstinence syndrome: An update. Res Rep Neonatol 2014;4:75–86. www.dovepress.com/clinical-presentation-and-management-of-neonatal-abstinence-syndrome-a-peer-reviewed-full-text-commodity-RRN (Accessed August three, 2017).
- Kocherlakota P. Neonatal abstinence syndrome. Pediatrics 2014;134(2):e547–61.
- Coyle MG, Ferguson A, Lagasse 50, Oh W, Lester B. Diluted tincture of opium (DTO) and phenobarbital versus DTO lone for neonatal opiate withdrawal in term infants. J Pediatr 2002;140(5):561–4.
- Ruwanpathirana R, Abdel-Latif ME, Burns L, et al. Prematurity reduces the severity and need for treatment of neonatal forbearance syndrome. Acta Paediatr 2015;104(5):e188–94.
- Hudak ML, Tan RC; Committee on Drugs; Committee on Fetus and Newborn; American Academy of Pediatrics. Neonatal drug withdrawal. Pediatrics 2012;129(2):e540–60.
- Newnam KM. The correct tool at the right time: Examining the evidence surrounding measurement of neonatal abstinence syndrome. Adv Neonatal Care 2014;fourteen(three):181–six.
- Finnegan LP, Kron RE, Connaughton JF, Emich JP. Assessment and treatment of abstinence in the infant of the drug-dependent mother. Int J Clin Pharmacol Biopharm 1975;12(1–ii):19–32.
- Asti Fifty, Magers JS, Keels E, Wispe J, McClead RE. A quality improvement project to reduce length of stay for neonatal abstinence syndrome. Pediatrics 2015;135(6):e1494–500.
- D'Apolito K, Finnegan LP. Assessing Signs and Symptoms of Neonatal Abstinence Using the Finnegan Scoring Tool: An Inter-observer Reliability Program. 2nd ed. Nashville, TN: NeoAdvances, 2010.
- Pulatie Chiliad. The legality of drug-testing procedures for pregnant women. Virtual Mentor 2008;10(1):41–4.
- Levy South, Siqueira LM, Ammerman SD et al.; Committee on Substance Abuse. Testing for drugs of abuse in children and adolescents. Pediatrics 2014;133(half dozen):e1798–1807.
- Gibbs J, Newson T, Williams J, Davidson DC. Naloxone take chances in babe of opioid abuser. Lancet 1989;2(8655):159–60.
- McKnight S, Coo H, Davies Chiliad et al. Rooming-in for infants at take chances of neonatal abstinence syndrome. Am J Perinatol 2016;33(5):495–501.
- MacMillan KDL, Rendon CP, Verma One thousand, Riblet N, Washer DB, Volpe Holmes A. Association of rooming-in with outcomes for neonatal abstinence syndrome: a systematic review and meta-analysis. JAMA Pediatr 2018 [Epub ahead of impress]. doi: 10.1001/jamapediatrics.2017.5195.
- Holmes AV, Atwood EC, Whalen B, et al. Rooming-in to treat neonatal forbearance syndrome: Improved family-centered care at lower cost. Pediatrics 2016;137(vi):e20152929.
- Hodgson ZG, Abrahams RR. A rooming-in program to mitigate the demand to treat for opiate withdrawal in the newborn. J Obstet Gynaecol Tin can 2012;34(5):475–81.
- Jansson LM, Velez One thousand, Harrow C. The opioid-exposed newborn: Assessment and pharmacologic management. J Opioid Manag 2009;5(1):47–55.
- Abdel-Latif ME, Pinner J, Clews Due south, Cooke F, Lui Thousand, Oei J. Furnishings of breast milk on the severity and outcome of neonatal forbearance syndrome among infants of drug-dependent mothers. Pediatrics 2006;117(six):e1163–9.
- Wachman EM, Byun J, Philipp BL. Breastfeeding rates among mothers of infants with neonatal forbearance syndrome. Breastfeed Med 2010;5(4):159–64.
- Maguire DJ, Rowe MA, Spring H, Elliott AF. Patterns of disruptive feeding behaviors in infants with neonatal abstinence syndrome. Adv Neonatal Care 2015;15(half dozen):429–39; quiz E1–ii.
- LaGasse LL, Messinger D, Lester BM, et al. Prenatal drug exposure and maternal and infant feeding behaviour. Arch Dis Child Fetal Neonatal Ed 2003;88(5):F391–9.
- White potato-Oikonen J, Brownlee Grand, Montelpare W, Gerlach K. The experiences of NICU nurses in caring for infants with neonatal abstinence syndrome. Neonatal Netw 2010;29(5):307–xiii.
- MacMullen NJ, Dulski LA, Blobaum P. Show-based interventions for neonatal abstinence syndrome. Pediatr Nurs 2014;40(4):165–72, 203.
- Bagley SM, Wachman EM, Holland E, Brogly SB. Review of the assessment and management of neonatal abstinence syndrome. Aficionado Sci Clin Pract 2014;9(1):19
- Kraft WK, Adeniyi-Jones SC, Chervoneva I et al. Buprenorphine for the handling of the neonatal abstinence syndrome. Due north Engl J Med 2017;376(24):2341–viii.
- Kraft WK, Stover MW, Davis JM. Neonatal forbearance syndrome: Pharmacologic strategies for the mother and infant. Semin Perinatol 2016;twoscore(iii):203–12.
- Streetz VN, Gildon BL, Thompson DF. Role of clonidine in neonatal abstinence syndrome: A systematic review. Ann Pharmacother 2016;50(4):301–10.
- Smirk CL, Bowman Eastward, Doyle LW, Kamlin CO. How long should infants at run a risk of drug withdrawal be monitored after nascency? J Paediatr Child Health 2014;50(5):352–5.
- Provincial Council for Maternal and Child Wellness. Neonatal Abstinence Syndrome (NAS) Clinical Practice Guidelines. July nineteen, 2011 (revised March 30, 2012): https://pqcnc-documents.s3.amazonaws.com/nas/nasresources/NASGuidelines.pdf (Accessed August 3, 2017).
- Smirk CL, Bowman Due east, Doyle LW, Kamlin O. Dwelling house-based detoxification for neonatal forbearance syndrome reduces length of hospital admission without prolonging treatment. Acta Paediatr 2014;103(6):601–four.
- McQueen K, Murphy-Oikonen J. Neonatal abstinence syndrome. Due north Engl J Med 2016;375(25):2468–79.
- Backes CH, Backes CR, Gardner D, Nankervis CA, Giannone PJ, Cordero L. Neonatal forbearance syndrome: Transitioning methadone-treated infants from an inpatient to an outpatient setting. J Perinatol 2012;32(6):425–30.
- Marcellus L, Loutit T, Cross S. A national survey of the nursing intendance of infants with prenatal substance exposure in Canadian NICUs. Adv Neonatal Intendance 2015;xv(5):336–44.
- Johnston A, Metayer J, Robinson Eastward. Management of Neonatal Opioid Withdrawal. Department 4 of Treatment of Opioid Dependence in Pregnancy: Vermont Guidelines. Vermont Children's Hospital at Fletcher Allen Health Care, 2010. http://contentmanager.med.uvm.edu/docs/default-source/vchip-documents/vchip_5neonatal_guidelines.pdf ?sfvrsn=2 (Accessed August 3, 2017).
Disclaimer: The recommendations in this position statement do not betoken an exclusive form of handling or procedure to be followed. Variations, taking into account individual circumstances, may be appropriate. Internet addresses are current at time of publication.
Source: https://cps.ca/documents/position/opioids-during-pregnancy
0 Response to "If Opiaters Used in First Trimester Pregnancy Only, Can Baby Have Withdrawal at Birth"
Post a Comment